
FDA Approved for the treatment of Depression
We were the first facility in North Carolina to offer the FDA approved NeuroStar TMS Therapy for the treatment of depression. The following information describes our TMS program. If you have any questions, please contact Sheila Mulford, TMS Coordinator, at 910-762-8400.
What is TMS?
TMS stands for “Transcranial Magnetic Stimulation”. NeuroStar® TMS Therapy is a medical procedure that works by delivering highly focused MRI-strength magnetic pulses to non-invasively stimulate the left side of the brain. The TMS treatment session is conducted using a small hand held device called a “treatment coil” that delivers pulsed magnetic fields. These magnetic fields are the same type and strength as those used in magnetic resonance imaging (MRI) machines. Patients being treated with TMS Therapy do not require anesthesia or sedation. They remain awake and alert and return to work or their daily routine after each treatment.
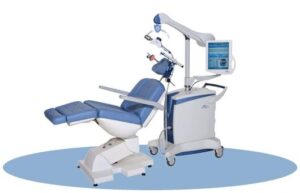
Benefits and advantages of TMS
- Non-invasive treatment
- Out-patient treatment
- Statistically and clinically significant improvement in depression symptoms (in open-label trial, 54% of patients responded and 33% remitted)
- No interruption to daily schedule
- No anesthesia
- No adverse effects on cognition
- Few or no side effects (most severe side effect is a mild headache, scalp discomfort). Less than 5% of patients discontinued treatment due to side effects.
- Safe and tolerable. More than 10,000 active treatments have been safely performed.
- Patients do NOT have to stop taking medication in order to receive TMS
- FDA approved
